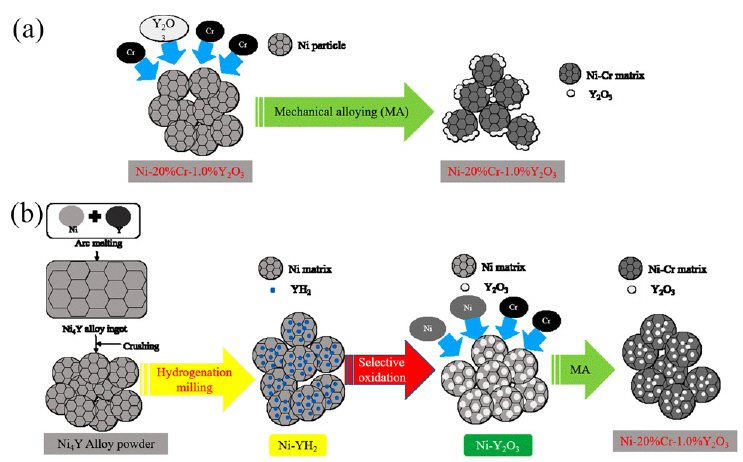
1. Introduction
Oxide dispersion-strengthened (ODS) Ni-based alloys are highly anticipated for high temperature applications. To prepare ODS Ni-based alloy for practical applications, oxide particles (such as Y2O3, Al2O3, and ThO2, etc.) are added to a nickel matrix [1]. In ODS Ni-based alloys, thermally stable oxide particles are evenly dispersed in a nickel matrix. The distribution of second-phase particles can hinder the movement of the dislocations and grain boundaries and reduce the diffusion rate of metal atoms, which can effectively improve the strength and high-temperature oxidation resistance of an alloy [2]. For high-performance ODS alloys, the oxide particles need to be refined and uniformly distributed in the metal matrix [3–6]. Because the strength of an ODS alloy is proportional to the inverse of the inter-particles distance, particle size refinement and the homogeneous distribution of particles is a very effective way to improve creep resistance [5]. ODS alloys are generally prepared using a powder metallurgical process, which involves the mechanical alloying (MA) of mixed or pre-alloyed powders with oxide, followed by pressure-assisted consolidation at high temperatures [7–9]. However, ODS alloys produced by conventional MA processes have some disadvantages, such as the rapid coarsening of oxide particles and metallic matrix grains with increasing temperatures, and a non-uniform distribution of oxide particles in the metallic matrix. When the adhesion between the oxide and the metal particles is poor, the oxide particles are less disturbed by the matrix and move faster during sintering, resulting in coarsening of the oxide particles and matrix grains, and uneven oxide dispersion [10,11]. These issues can be largely overcome by improving adhesion between the oxide and the metallic particles of the raw material powder used for sintering. Previous studies on way to improve the adhesion problem have involved the use of a raw material powder composed of the oxide-embedded particles for sintering [12]. In the present study, to prepare a Ni–20wt.% Cr–1.2wt.% Y2O3 alloy powder composed of metallic particles with embedded oxide for sintering, Ni–YH2 composite particles were first synthesized by ball milling a Ni4Y alloy powder under pressurized H2 gas. The Ni–YH2 particles were then selectively oxidized to produce Ni particles with embedded Y2O3. To confirm the composition, Ni and Cr powders were added to the Ni-Y2O3 composite prepared by the aforementioned process, and then mechanically alloyed for 25 h under an inert gas atmosphere. For comparison, alloy powders with the same composition were prepared by conventional MA in an inert gas atmosphere. The two samples were consolidated using spark plasma sintering. The microstructures of the Ni–Cr–Y2O3 alloy powder prepared using Ni particles with embedded Y2O3 were observed and compared with those of a conventionally alloyed Ni–Cr–Y2O3 powder. In addition, the mechanical properties and the strengthening mechanism of the sintered alloys were investigated.
2. Materials and Methods
First, Ni4Y ingots were prepared by the arc melting of Ni (purity: 99.95%, Avention) and Y (purity: 99.9%, Alfa Aesar). The Ni4Y ingot was then crushed and sieved to a particle size of less than 120 μm. The Ni4Y alloy powder was hydrogenated into a Ni–YH2 composite using planetary high-energy ball milling under a pressured H2 gas atmosphere for 25 h. During milling, the H2 gas pressure in the vial was filled to 3 atm every 5 hours. About 20 g of powder was put into a stainless steel vial along with stainless steel balls (diameter 10 mm) and milled; the powder ball weight ratio was 1:10. The disc and vial rotating speeds were 600 and 320 rpm, respectively. The Ni–YH2 powder prepared by mechanical hydrogenation was oxidized at 600 °C for 3 h. Then, the Ni phase oxidized in the oxidation process was reduced at the same temperature for 1 hour using H2 gas. To confirm the composition of the Ni–20wt.%Cr–1.2wt.% Y2O3 alloy, Ni and Cr powders were added to a Ni–Y2O3 composite prepared using the above process. The mixed powder was alloyed by planetary ball milling in Ar gas. In the conventional mechanical alloying method used for comparison, a mixed powder of Ni (purity 99.9%, 3–7 µm, Alfa Aesar), Cr (purity 99%, under 254 µm, Alfa Aesar) and Y2O3 (purity 99.99%, 0.9–1.6 µm, Alfa Aesar) was alloyed by planetary ball milling in an Ar gas. Prior to the mechanical alloying, stearic acid (purity 99.5%, Samchun) of 1 wt.%, , was added to both mixed powders as a process control agent (PCA). To investigate the changes in the crystal structure of the powder, a sample was taken every 5 hours and the sample was rested for 30 minutes. The total milling time was 25 hours. Both mechanically alloyed powders were sintered using a spark plasma sintering (SPS) system. A graphite die with an inner diameter of 10.5 mm and an outer diameter of 40 mm was used. All SPS experiments were conducted under vacuum (under 3 × 10-3 torr). The sintering was performed at 850, 900, 950, and 1,000 °C, and the holding time was 1 min each. The heating rate and applied pressure were 300 °C/min and 80 MPa. The densities of the specimens were measured using the Archimedes method. The crystal structures of the powders were analyzed by X-ray diffraction (XRD) with a CuKα source (D-Max 2500, Rigaku). The microstructure of the powder particles was observed by STEM (JEM-ARM 200F, JEOL), and analysis of the results was performed using fast Fourier transformation (FFT). Electron backscatter diffraction (EBSD) images were obtained using field emission electron microscopy (FE-SEM; S-4800, Hitachi). Hardness tests were conducted using a Vickers hardness (Hv) tester. The compressive test was performed using a universal testing machine (8801, Instron) with a strain rate of 10-3 s-1. The diameter-to-height ratio of the tested sample was 1:2 (the diameter and height were 5 and 10 mm, respectively).
3. Results and Discussion
3.1 Preparation of Ni particles with embedded Y2O3 grains
In this study, a new fabrication process was used to prepared Ni-based ODS alloys. The new process involved the preparation of an Ni-Y alloy powder by arc melting and crushing an alloy ingot. The two metallic phases in the Ni4Y alloy were converted into Ni + YH2 phases by hydrogenation milling, and the partially hydrogenated particles were selectively oxidized to Ni + Y2O3 phases. In addition, to ensure the desired composition (Ni–20wt.%Cr–1.2wt.% Y2O3), Ni and Cr powders were added to the Ni particles with embedded Y2O3, and then the mixed powder was mechanically alloyed.
Figure 1 shows the XRD pattern of (a) the raw alloy, (b) hydrogenated powder milled under pressured hydrogen gas, and (c) the oxidized and reduced hydrogenated powders. The Ni4Y ingot consisted of Ni3Y and Ni5Y phases. When the alloy powder was milled under pressurized H2 gas for 25 h, only the Y element of the alloy was converted into the YH2 phase. That is, the Ni3Y and Ni5Y phases in the ingot were reformed into Ni and YH2 phases, as shown in Figure 1(b). When the Ni–YH2 powder was oxidized and reduced at 600 °C, only the YH2 phase was oxidized to Y2O3, as shown in Figure 1(c). For convenience, in the following the Ni–Cr–Y2O3 composite prepared using the new process is referred to as NCY-h, and the Ni–Cr–Y2O3 composite powder prepared using the conventional MA process is referred to as NCY-c.
Figure 2 shows the XRD pattern of (a) the NCY-c samples prepared using the conventional MA process and (b) the NCY-h powder prepared using the new process introduced in this study. The diffraction peaks of Cr and Y2O3 in the NCY-c and NCY-h powders were almost eliminated by the MA effect. The impact energy generated by ball milling generally results in a reduction in crystallite size and an accumulation of lattice strain [13]. As a results, only a single phase of fccNi remained in both powders. The peak intensities of the fccNi phase in NCY-h were slightly lower than those of the NCY-c powder. The microstructures of the NCY-c and NCY-h powders were observed using STEM. The results were analyzed using fast Fourier transformation (FFT). As shown in Figure 3(a), Y2O3 particles, approximately 4–5 nm in diameter, exist only on the surface of the matrix Ni(Cr) particle. Since the metallic Ni(Cr) and Y2O3 particles cannot react chemically, Y2O3 particles, which are fragile in the MA process, are located on the surface of the metallic Ni(Cr) particles. Unlike the NCY-c particles, the Y2O3 grains in the NCY-h sample were placed inside the Ni(Cr) particles, as shown in Figure 3(b). It appears that the decomposition and conversion reactions of the phases occurring during the new process, such as hydrogenation, oxidation, and reduction, occurred only within the particle. A schematic of the conventional MA process and the new process applied in this study for the fabrication of the Ni-based ODS alloy is shown in Figure 4. The conventional MA process was carried out by mechanical alloying of the powder mixed with the metals and oxides components of the desired composition (Ni–20wt.% Cr–1.2wt.%Y2O3), as shown in Figure 4(a). The new process consisted of the preparation of a Ni–Y alloy powder, recomposition of the two metallic phases in the Ni–Y alloy into Ni–YH2 particles through hydrogenation milling, the selective oxidation of Ni–YH2 into Ni–Y2O3, and then MA of the powder with the desired composition (Ni–20wt.%Cr–1.2wt.%Y2O3), as shown in Figure 4(b). This new process was very effective in manufacturing the metallic Ni(Cr) particles with embedded Y2O3 grains, which is needed to improve adhesion between the oxide particles and the metallic matrices.
3.2 Microstructures of the Sintered alloys
The NCY-c and NCY-h powders were consolidated by the SPS machine at 850, 900, 950, and 1,000 °C, respectively. Table 1 shows the calculated relative densities. All of the sintered specimens showed relative densities higher than 99.7%, regardless of the sintering temperature. The high relative density, even at low temperatures, is believed to be due to the fabrication of finer and more active particles depending on the milling conditions, and the higher pressure and heating rate applied during sintering with the SPS machine.
Figure 5 shows the TEM images and Y2O3 particle distribution of the (a) NCY-c and (b) NCY-h sintered at 850 °C, and the (c) NCY-c and (d) NCY-h sintered at 1000 °C. The Y2O3 grains in the NCY-c sample were located on the surface of the metallic Ni(Cr) particles, while in the NCY-h sample the Ni(Cr) particles were embedded in the Y2O3, as shown in Figure 3. When both powders were heated and sintered, the Y2O3 particles in the NCY-c sample were easily concentrated and grew into the boundary between the Ni(Cr) particles. However, the Y2O3 grains in the NCY-h sample were contained inside the Ni(Cr) particles, so the growth was delayed during the sintering process. These results indicate that the new process applied in this study was effective at inhibiting the growth of Y2O3 particles during sintering. Table 2 shows the mean particle size, the inter-particle distance of Y2O3 and standard deviation in particle sizes. The results are summarized in Figure 5. The mean inter-particle distance was calculated using equation (1) [14]:
where λ is the mean particle separation (in nm), r is the mean particle radius, and f is the volume fraction of the dispersion particles. The average particle diameter, inter-particle distance and standard deviation of the two samples increased with increasing temperature. The particle diameter and inter-particle distance in NCY-h were smaller than those in NCY-c. On the other hand, the standard deviation of their particle diameters were similar at each temperature.
Figure 6 shows the EBSD micrographs of NCY-c sintered at (a) 850 °C, (b) 900 °C, (c) 950 °C, and (d) 1,000 °C, and NCY-h sintered at (e) 850 °C, (f) 900 °C, (g) 950 °C, and (h) 1,000 °C. The grain sizes of the matrix in the NCY-h and NCY-c samples, obtained by analyzing the results in Figure 6, are summarized in Table 3. The grain size of the matrix in the NCY-h sample was smaller than that in the NCY-c sample. The finer Y2O3 particles distributed in the NCY-h sample obstructed the migration of the grain boundaries in the matrix. Although both samples had the same amount of oxide, the refinement of the oxide in the NCY-h sample increased the number of obstacles, leading to the refinement of the matrix grains.
3.3 Mechanical Properties and Strengthening Mechanism
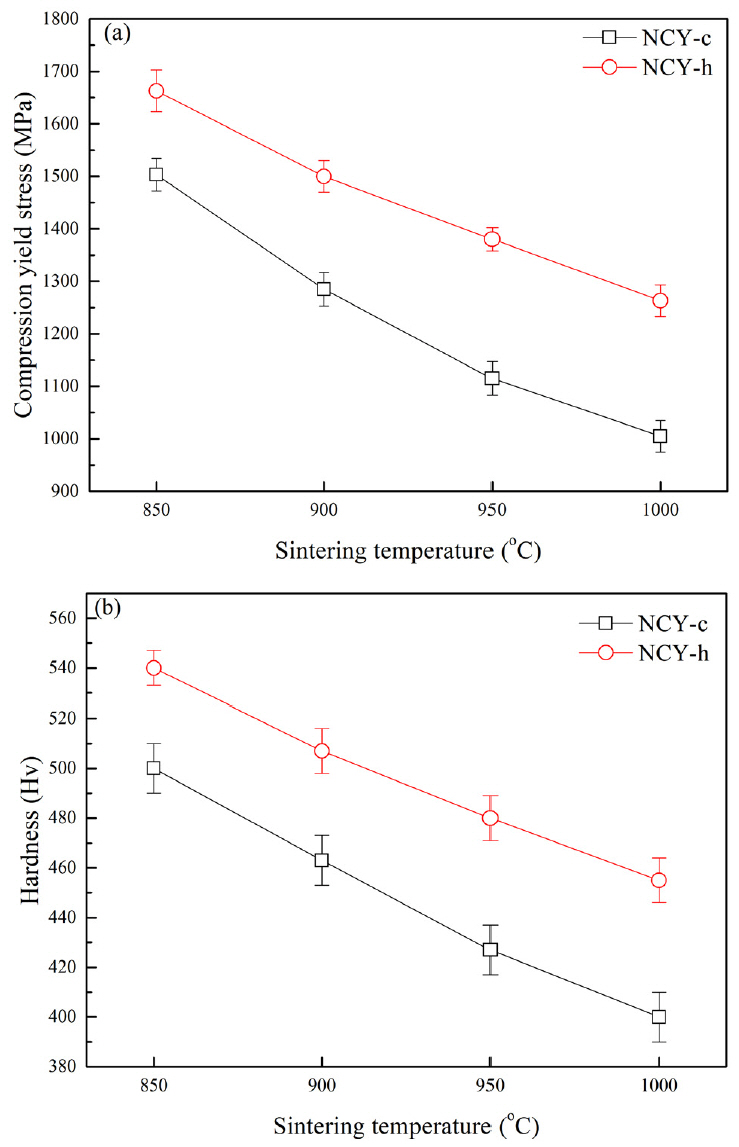
Figure 7 shows the (a) compression yield strength and (b) Vickers hardness of the sintered NCY-c and NCY-h samples. The compression yield strengths of the NCY-c and NCY-h samples reached their highest values at 850 °C and decreased with increasing sintering temperature. The compression yield strength and Vickers hardness of the NCY-h sample were higher than those of the NCY-c sample at all temperatures. The NCY-h sample was more reinforced compared to the NCY-c sample, because of the finer oxide particles and smaller matrix grains obtained with the new process. The strengthening of the ODS alloy can be attributed mainly to a combination of the Hall–Petch (owing to a matrix grain refinement) and the Orowan (owing to precipitation or distribution of the oxide) mechanisms. The total strengthening was estimated through a simple linear addition of all contributing mechanisms, assuming that the strengthening mechanisms operated independently of each other [15]. Hence, σY can be estimated using equation (2):
where σY is the estimated yield strength; σ0 is the Peierls–Nabarro stress, or lattice-friction stress, and is negligible; Δσss is the solid solution strengthening contribution; ΔσD is the dislocation strengthening contribution; ΔσGB is the grain boundary strengthening contribution; and ΔσOro is the Orowan, or dispersion mechanism contribution. σ0 (lattice friction stress), for a pure fcc crystal structure at room temperature, is 6–8 MPa [16].
The solid solution strengthening from multiple alloying elements in an Ni alloy has been investigated [17], and the strengthening can be expressed using the following relation:
where Δσss is the solid solution contribution, ki is the strengthening constant for solute i, ci is the concentration of solute i, and n is taken as 0.5 herein. For the Ni–Cr alloy, the value of k is 337 MPa at fraction-1/2 [18]. The strengthening due to the Ni-20wt.%Cr solid solution was 158 MPa, assuming that the Y2O3 particles did not dissolve in the matrix [15].
The strengthening contribution by dislocation can be calculated using the Bailey–Hirsch equation [19]:
Where the α value of the dislocation strengthening coefficient is 0.25 [20]. M (Taylor factor), G (shear modulus), and b (Burgers vector) were taken as 3, 82 GPa, and 0.25 nm, respectively [21]. The dislocation density was calculated using XRD analysis. The dislocation densities of the NCY-c and NCY-h samples sintered at 1000 °C were 3.7 × 1014 m−2 and 1.8 × 1014 m−2, respectively. This indicates that the Y2O3 particles in the NCY-h acted more strongly as obstacles to dislocation motion.
The grain boundary strengthening contribution (Hall–Petch relationship) can be evaluated with the following equation [22]:
where k is the Hall-Petch coefficient, whose value is 5538 MPa·nm1/2 [23], and D is the mean size of grains, based on the results shown in Table 1. The contributions to grain boundary strengthening in the NCY-c and NCY-h samples sintered at 1000 °C were 345.1 MPa and 177.1 MPa, respectively.
The strengthening contribution attributed to the Orowan mechanism can be evaluated using the following equation [24]:
where M is the Taylor factor, r is the average particle radius, λ is the mean particle distance, and v is the Poisson ratio. The values of r and λ are incorporated in the results from Table 1. The strengthening contributed by the oxide particles depends on r and λ due to pinning effects. Therefore, a reduction in λ and the homogeneous distribution of the oxide particles could intrinsically improve the strength of the samples [20,24]. Table 4 showed a summary of the contributions of the different strengthening mechanisms in the NCY-c and NCY-h samples sintered at 1000 °C and a comparison of the estimated yield strengths with the experimentally determined values. The calculated yield strength was in close agreement with the experimental values. The values of ΔσD, ΔσGB, and ΔσOro for the NCY-h prepared by the new process applied in this study were higher than those of NCY-c. The NCY-h sample had oxide particles of a finer size and closer distance than the NCY-c sample, as shown in Table 2. The dispersion strengthening (Orowan mechanism) was attributed to refinement of the oxide particles. In addition, the fine oxide particles promoted the refinement of the matrix through a pinning effect. Based on the results in Figure 6 and Table 3, the matrix particles of the NCY-h sample were relatively more refined at all sintering temperatures. In addition, NCY-h had the same oxide volume as NCY-c, but the oxide particles in NCY-h were finer, reducing the distance between the particles, which promoted the accumulation of dislocations in the matrix, thereby resulting in strengthening. This indicates the refinement of oxide particles in the NCY-h sample was responsible for the enhancement of the mechanical properties.
4. Conclusion
An NCY-h composite powder consisting of Ni(Cr) metallic particles with embedded Y2O3 grains was prepared using a new process introduced in this study. For comparison, an NCY-c powder was prepared using a conventional MA method. Samples of the two powders were sintered using the SPS method at temperatures 850, 900, 950, and 1,000 °C. The relative densities of all sintered samples were higher than 99.7%. Regardless of the sintering temperature, the particle size and inter-particle distance in the NCY-h sintered samples were smaller than those of NCY-c. Mechanical properties such as the compressive yield strength and Vickers hardness of the two sintered metals decreased with increasing sintering temperature. On the other hand, the compressive yield strength and Vickers hardness of the sintered NCY-h showed higher values compared to NCY-c at all sintering temperatures. The enhancement of the mechanical properties of the NCY-h sample was attributed to the refinement of oxide particles fabricated by the new process.